Whale Imaging, Inc., creator of the G-Arm and Sigma Series ultrasound medical imaging systems, announced FDA 510(k) clearance of the G-Arm B6 Duo.
The B6 Duo is the next generation of the G-Arm with improvements including axial tilt and greater table access.
“We’re thrilled to be launching this sooner than our original intended date,” says Laurence Heron, SVP of Sales and Marketing at Whale. “We feel that getting the B6 Duo into our customer’s OR will really revolutionize surgical imaging for orthopedic, spine and neuro surgeons as well as pain practitioners.”
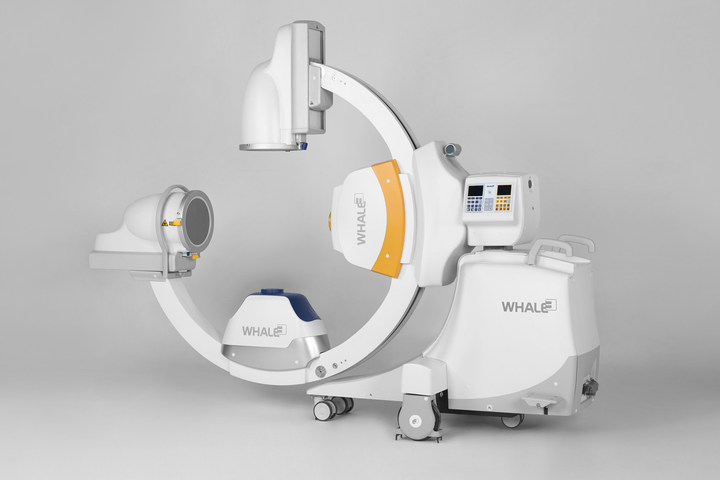
Multiscan Imaging System (Credit: Whale Imaging Inc.)
The Whale Imaging research and development team’s mission was to help the user improve accuracy while reducing time, infection and radiation. Engineers at Whale designed the B6 Duo to be compatible with all surgical tables, and added features including axial tilt. Whale’s ability to fire two x-ray beams asynchronously provides the customer with bi-lateral video and images, giving the user a more comprehensive picture than a traditional C-arm.
G-Arm utilizes X-Beam technology as well as iC-Clear algorithms to provide the best image possible, using the smallest dose of radiation.
“Within a few years, we expect the B6 Duo to be a common fixture in orthopedics and spine departments across the US,” says Phil Posa, VP of the G-Arm Division. “It has many advantages over C-Arms for various procedures, so why add another C-Arm to your inventory when a G-Arm can accomplish so much more?”
The G-Arm B6 Duo is now available for sale from Whale Imaging, and is FDA 501(k) approved.




