In a recent study, a Yale Cancer Center team compared the performance of the four available PD-L1 assay tests. They found that one of the assays failed to reveal comparable levels of PD-L1, a tumor-promoting protein, while three others revealed comparable levels. The findings were presented at the International Association for the Study of Lung Cancer (IASLC) 2016 Chicago Multidisciplinary Symposium in Thoracic Oncology.
PD-L1 assays are used to test expression of the PD-L1 protein on a patient’s tumor to provide customized treatment options for drugs that may be more effective than chemotherapy in lung cancer.
Yale team compares effectiveness of the four PD-L1 tests. (Credit: Yale Cancer Center)
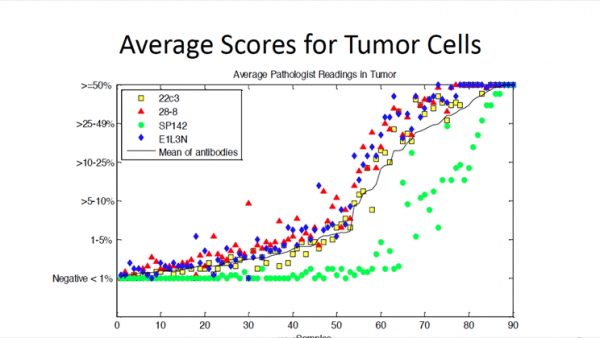
The research team reviewed 90 surgically resected non-small cell lung cancer (NSCLC) cases (stages I-III) and sent a sample of each to four facilities for staining. A group of 13 pathologists at seven institutions then reviewed the samples, using four different assays on each case, and scored them using a unified scoring system. A comprehensive statistical analysis was then performed on the scores collected.
Currently, there are four drugs with four PD-L1 assays (22c3, 28-8, E1L3N, and SP142) available; but only the 22c3 test is required by the FDA for prescription of a targeted anti-PD-1 drug (pembrolizumab), while the others are not yet required for prescription of other PD-1 axis drugs.
Analysis revealed that one of the assays, SP142, systematically returned statistically lower levels of PD-L1 expression than the other three. This was true in both tumor and immune cells using any test. The remaining three assays available (28-8, E1L3N, and 22c3) showed no significant difference between them, according to researchers.
“Our data shows that the SP142 assay shows significantly lower levels of PD-L1 expression. This observation may limit the use of this assay in PD-L1 testing moving forward,” said David L. Rimm, M.D., Ph.D., first author on the study and a professor of pathology and of medicine (Medical Oncology), Yale School of Medicine. “However, the other three assays seem equivalent, which is good news for the future when other PD-1 axis drugs with assay-specific diagnostics gain FDA approval.”
Bristol Myers Squibb with the National Comprehensive Cancer Network (NCCN) Oncology Research Program sponsored this study.
(Source: Newswise)
