CytoSorbents (NASDAQ: CTSO) said it has won another round of funding from the U.S. government to commercialize its HemoDefend red blood cell (RBC) transfusion filter.
CytoSorbents (NASDAQ: CTSO) said it has won another round of funding from the U.S. government to commercialize its HemoDefend red blood cell (RBC) transfusion filter.
The critical-care immunotherapy company, based in Monmouth Junction, N.J., specializes in blood purification. The National Heart, Lung, and Blood Institute (NHLBI), a division of the National Institutes of Health, granted the three-year, Phase IIB Bridge Small Business Innovation Research award of up to $3 million.
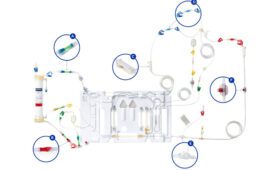
The HemoDefend-RBC filter is designed to improve the safety and quality of the blood supply by reducing non-infectious contaminants in packed red blood cells such as potassium, free hemoglobin, bioactive lipids, cytokines and antibodies. These contaminants can cause problematic and potentially life-threatening transfusion reactions. More than 100 million RBC units are transfused worldwide each year, and according to the American Red Cross, someone in the U.S. needs blood every 2 seconds.
NHLBI and U.S. Special Operations Command (USSOCOM), previously awarded the company $1.7 million in SBIR Phase I and II funding for HemoDefend, according to a company statement.
“In this next phase, NHLBI will match company funds dollar-for-dollar up to approximately $1 million each year for three years, for total funding of approximately $3 million, subject to the availability of funds and satisfactory progress of the project,” said CytoSorbents CEO Dr. Phillip Chan in a company statement. “This funding will be used to help finance the costs of the HemoDefend pRBC pivotal clinical trial that is expected to begin in Q1 2019 and support U.S. FDA regulatory approval, as well as initial commercialization activities, including manufacturing scale-up. We thank NHLBI for their continued support.”
CytoSorbents’ purification technologies are based on biocompatible, highly porous polymer beads that can actively remove toxic substances from blood and other bodily fluids by pore capture and surface adsorption. The federal support has allowed CytoSorbents to rapidly design, test, optimize, and validate the HemoDefend RBC filter in internal and external studies, according to Maryann Gruda, the company’s director of biology.
“This process highlights the flexibility of our bead-based blood purification platform and the deep expertise of translational product development at our company,” Gruda said in the statement. “Following our initial pre-submission discussions with the FDA that were facilitated by NHLBI, we look forward to potentially obtaining investigational device exemption protocol approval, successfully executing the pivotal study, and making the HemoDefend RBC filter available to patients in need in the U.S. and around the world.”
CytoSorbents’ technologies have received non-dilutive grant, contract, and other funding of approximately $25 million from DARPA, the U.S. Army, the U.S. Department of Health and Human Services, the NIH, the NHLBI, USSOCOM, and others.
The HemoDefend RBC filter is not approved in the U.S. or elsewhere.