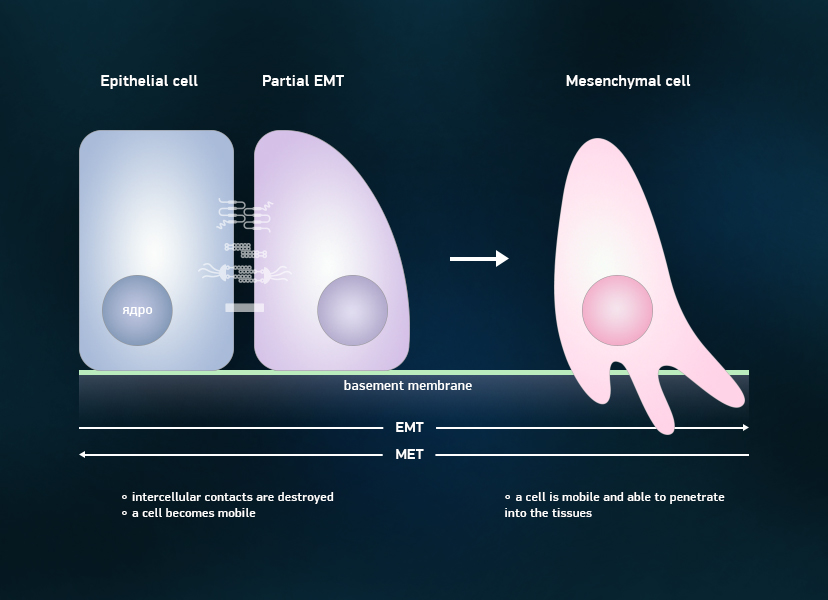
The figure shows changes in a cell shape and a number of other changes — for example, those in internal cell structure, plus the contacts between cells. In addition, on the right side of the figure, we indicated such a feature of mesenchymal cells as their ability to penetrate the extracellular matrix (or, more simply, the extracellular environment). The increased mobility of these cells contributes to the spread of cancer. (Credit: Moscow Institute of Physics and Technology)
MIPT scientists together with their colleagues from St. Petersburg and Israel have analyzed more than 500 previously published scientific articles and proposed their own approach to the choice of methods used for the treatment of one of the most common cancers. Details are published in the review of the International Journal of Cancer.
While making their choice, the researchers proposed to use clinical data on the concentration of certain proteins (biomarkers). With this information, mathematical models developed by this team allow making a choice between the conservative treatment (without surgery) and radical (surgical) intervention. Moreover, the authors believe that this approach will allow to select the optimal treatment for each patient.
Bladder cancer is one of the most common cancers; statistics show that one in 26 men and one in 90 women are susceptible to this form of cancer. In Russia, the number of cases is estimated at 12,500 per year, and therefore the problem of accurate diagnosis of the cancer stage and proper treatment is important both from a scientific and clinical point of view.
With proper diagnosis and timely detection, non-invasive bladder cancer can be successfully treated. The tumor practically always begins to grow in the layer of cells that lines the bladder from inside—the epithelium—and if the tumor does not have time to affect the muscle layer, then it is possible to remove it with minimal risk of recurrence. However, if the tumor has penetrated into the muscle of the bladder, then the cancer is called muscle-invasive; the latter is much more active in producing metastasis, and chances of full recovery are sharply decreased. Nowadays, the five-year survival rate for muscle-invasive bladder cancer is 30 to 50 percent—these figures indicate the number of patients surviving for five years after the discovery of such a tumor.
“In this work we correlate the latest clinical, molecular and cellular data associated with muscle-invasive bladder cancer. We use this data for developing a mathematical model of the disease. The ultimate goals of our work are, firstly, the identification of biomarkers and specific clinical parameters by means of which it is possible to accurately determine the stage of this aggressive form of cancer; secondly—the development of individual strategies for its treatment in each particular case,” says Alexander Kiselev, a professor of MIPT and lead author of the new article.
Bladder cancer tumors have different biochemical and cellular characteristics, different genetic and epigenetic profiles, and all this variation can even be found in a single patient. The particular mutations that occurred in the DNA of a cancer cell affect how the tumor responds to therapy. Determining the exact type of tumor (its profile) can significantly improve the chances of successful treatment and, at the same time, minimize side effects.
To determine the genetic profile means to establish the sequence of nucleotide pairs in the DNA. This sequence determines which molecules of the protein and RNA (ribonucleic acid) will be synthesized. The epigenetic profile refers to a list of active and “silent” genes: if a DNA region or chromosomal protein is modified, then a part of genes will cease to be used.
The molecular basis of cancer
Normal cells are transformed into cancer cells due to a series of failures at the genetic and molecular levels. A key role in this process is played by the so-called epithelial-mesenchymal transition—epithelial cells, which form a layer on the tissue surface, lose their normal structure and fail to respond to chemical signals, which should limit their division and / or cause their self-destruction (apoptosis). This transition is caused by a number of factors—in particular, by synthesis of the increased amount of certain proteins.
The process of epithelial-mesenchymal transition (EMT) resembles the process of the creation of normal cellsworking in reverse—those initially obtained from stem cells that are characterized by a broader specialization, and can divide a much greater number of times. But if the “right” stem cell does not form a tumor and is necessary for tissue regeneration, the EMT process can result in the occurrence of cancer—mesenchymal cells stop responding to signals coming from the surrounding tissues and begin grow uncontrollably. Proteins that are characteristic for this phase—such as the epithelial growth factor or proteins of the snail family—can help detect the disease in the very beginning.
Another protein that plays a critical role in the diagnosis and treatment of cancer is protein p53. It is known that this protein prevents the transformation of a healthy cell into a cancer cell by triggering a process of self-destruction when DNA is damaged and various stresses take place (in a biochemical, rather than a psychological, sense). This mechanism effectively protects the body from malignant tumors, but it cannot cope with those mutations, which occur within the gene coding protein p53. Such problems are known for many types of cancer; they allow detection of the disease, but do not give the information necessary for the correct choice of treatment.
Finally, an important sign of the aggressive muscle-invasive bladder cancer is a change in the concentration of protein Shh. In the developing organism, this protein is essential for differentiation of embryonic cells—but its aberrant expression may be associated with the development of invasive cancer.
The letters Shh stand for the words “sonic hedgehog”, which refer to the history of discovery of the gene bearing this name. First, scientists have discovered a gene which mutation covered the experimental Drosophila flies with something resembling hedgehog needles—they coded this as “hh”, short for “hedgehog.” The witty geneticists added to this description the word “Sonic” in honor of the fictional hedgehog—a character from the game Sonic the Hedgehog. Then they discovered genes “desert hedgehog” and “Indian hedgehog.”
From the knowledge of proteins to diagnosis
After analyzing the data, the authors proposed an approach to the treatment of bladder cancer as follows: First, all patients are tested for selected cancer development markers—proteins of the Snail family, Shh proteins and others. For this purpose, they perform a biopsy: A small piece of the tumor is pinched off, then that piece is treated with alcohol or formalin—substances that protect it from decomposition. The sample is embedded in paraffin, and then, with the help of a special knife called a microtome, the sample is cut into thin slices.
In order to reveal the tumor structure, the slices are “developed” by using reagents containing the modified antibodies—proteins capable of binding to specific parts of tissue—antigens. Antigen-antibody reactions occur in our bodies every day—immunity is based on these reactions. When, for example, germs enter the body, the specialized cells of the immune system synthesize antibodies that bind to germs and prevent them from spreading. Immunohistochemical studies are based on the same principle. If the antigen is present in the sample, then it reacts with the antibody. This process can be compared to fingerprinting—the surface appears clean, but after treatment with a special powder it shows the pattern of papillary lines.
In order to see antibodies, fluorescent dyes are added. Studying the treated slices under a microscope with a set of filters, scientists can detect the presence of cancer cells; and under high magnification, they are able to observe in detail the location of the studied proteins inside the cells.
To find the important tumor development markers, the tissue sample is also divided into many pieces that are spread over the microscope slide. Each of these pieces is exposed to specific antibodies. Therefore, from a single piece of tissue, scientists can collect data on many biomolecules. In the end, this allows them to determine the stage of the tumor development, and predict its aggressiveness and invasiveness. At the same time, the multiplicity of samples on one piece of glass makes it possible to select some individual markers, taking into account the patient’s medical history and his/her associated illnesses.
The obtained qualitative and quantitative data on the expression of specific biological molecules (marker panels) will be used in the mathematical model proposed by the authors, in order to:
- Assess and, if necessary, adjust the initial clinical diagnosis;
- Optimize the chosen method of treatment, the nature and dose of drugs;
- Predict the potential side-effects for each particular patient.
After the completion of treatment, it is necessary to conduct further tests using the selected marker panels in order to evaluate the results. The authors understand that depending on the results, a radical change of the course of treatment might be necessary. Such an approach to the management of medical history will provide the necessary clinical data for a large number of patients—that should help attending physicians improve the diagnosis process and the process of selection of individual treatment for each patient. At a time when every day counts, this is a real opportunity to prolong lives of many patients.
