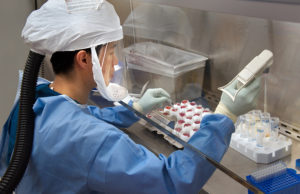
4. Tackling the shortage of tests
[Image courtesy of CDC]
Roche (SWX: RO, ROG) says it could eventually have millions of tests available per month to run on its high-volume Cobas 6800/8800 system. Thermo Fisher Scientific (NYSE:TMO) has 1.5 million tests available to ship, and Hologic (NSDQ:HOLX) expects to ship tens of thousands of tests this month. Abbott (NYSE:ABT) will immediately ship 150,000 of its RealTime SARS-CoV-2 EUA tests to existing U.S customers.
Two companies — Everlywell and Nurx — plan to start offering at-home COVID-19 tests in coming days.
Medical experts told The Wall Street Journal that the emergency use authorization should ease the shortage of tests, but at the potential cost of the tests being less reliable.




![A photo of the Medtronic GI Genius ColonPro polyp detection system flagging a potential sign of colon cancer during a colonoscopy. [Photo courtesy of Medtronic]](https://www.medicaldesignandoutsourcing.com/wp-content/uploads/2024/04/Medtronic-GI-Genius-doctors-268x170.jpg)