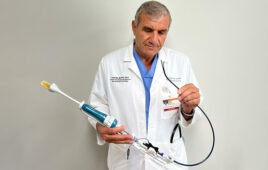
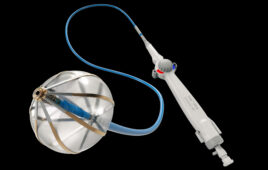
Integer’s new radial access introducer kit for interventional cardiology and peripheral vascular procedures has won FDA clearance and the CE Mark.
Integer’s new radial access introducer kit for interventional cardiology and peripheral vascular procedures has won FDA clearance and the CE Mark.
RadialSeal was engineered for optimal device compatibility and ease of insertion, the company said in a statement. Its thin-walled sheath design minimizes outer diameter while maximizing inner diameter, according to Payman Khales, president of Integer’s cardiovascular division. The introducer also offers easy sheath insertion through smooth transitions and hydrophilic coating, Khales added.
The device is Frisco, Texas-based Integer’s response to the limited number of viable offerings in the thin-wall introducer segment of the growing radial access market, he said.
“We set out to build upon our proven hemostatic introducer and guidewire technologies to develop a robust radial introducer portfolio ranging from 4F to 6F to address the needs of the interventional cardiologists,” said Andrew Senn, vice president of product solutions. “The thin wall sheath design increases radial access options, especially for patients with smaller arteries, while the smooth transitions and tip, combined with a hydrophilic coating, lessen friction to potentially reduce the risk for vessel trauma and spasm.”
The RadialSeal Introducer Kit is founded on Integer’s core technology platforms in guidewires and hemostatic introducers, adding to its portfolio of access products available for distribution by medical device companies in the interventional cardiology and peripheral vascular space.