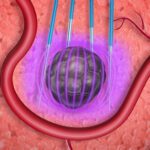
The Abbott Volt aims to beat first-generation pulsed field ablation (PFA) systems with its balloon-in-basket design and other features.

The Abbott Volt pulsed field ablation (PFA) catheter features a balloon-in-basket design and eight electrode splines. [Image courtesy of Abbott]
The Volt cardiac ablation catheter has a balloon, something you won’t see on Medtronic’s PulseSelect PFA catheter — the first of its kind approved by the FDA — or the Farawave catheter in Boston Scientific’s Farapulse PFA system.
The Abbott Volt’s balloon-in-basket is designed to support the efficient deployment of energy into the tissue during cardiac ablation to treat atrial fibrillation (AFib). That energy creates lesions, scarring the heart tissue to block the irregular electrical signals that disrupt the heart’s normal rhythm.
Abbott said the catheter’s design improves the accuracy, quality and efficiency of ablation to minimize the number of applications needed to treat a patient, as well as potential side effects.
“We have data showing if you use a balloon versus a basket without the balloon it makes a difference [in] lesion depths by 20% to 30%,” said Dr. Christopher Piorkowski, the chief medical officer of Abbott’s electrophysiology division, in an interview with Medical Design & Outsourcing.
To further maximize efficient energy transfer into the heart tissue, Abbott designed the device with electrodes that only face outward and splines that are flat, not round.
Based on Abbott’s bench and animal testing data, the Volt design can drive chronic lesion depths of 6 to 8 mm, Piorkowski said, calling those depths “very sufficient for the left atrium and pulmonary vein isolation.”
At the same time, the balloon acts as an insulator for the blood inside the patient’s beating heart, reducing thermal effects such as bubble formation. Piorkowski said the Volt also uses less voltage for ablation than competing PFA devices, reducing the risk of thermal effects. (He declined to say how much voltage Abbott’s system uses or to quantify the difference.)
The balloon helps stabilize the basket inside the pulmonary vein, where the device’s round shape allows a physician to place the catheter at the ablation target site, apply energy, and rotate the device slightly to achieve a spline offset for a second energy application. In tests, physicians are using three to four applications per treatment.
“That is way below everything that is currently being done with PFA in the clinical field,” he said.
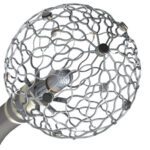
Measuring tissue contact during PFA

The Abbott Volt PFA System can tell a physician whether the catheter’s electrodes are in good contact with heart tissue to maximize ablation effectiveness. [Image courtesy of Abbott]
Abbott engineers took an impedance-based measurement that the company invented for previous ablation catheters and applied it to each of the Volt’s electrodes.
“We immediately saw the benefit in bench testing [and] also in the clinical field,” Piorkowski said. “In PFA, we have enabled the system to perform such contact assessment not on a single electrode, but on all eight splines of the device.”
What’s next for the Abbott Volt PFA system

Abbott Electrophysiology Chief Medical Officer Dr. Christopher Piorkowski [Photo courtesy of Abbott]
Because PFA is relatively new, the FDA and other regulators are learning about it along with the medtech industry, taking what they learn from publications, conferences and presentations and bringing their questions to device developers like Abbott during clinical trial design.
“Bubble formation was something in the beginning of the PFA era that was little talked about,” Piorkowski said. “When we designed our clinical trial — and we have a lot of pre-sub meetings, for instance, with FDA where we talk about the trial design and what they feel is important and how we should address it — this topic of bubble formation came up and we had to address it in preclinical testing and also in the trial design.”
The IDE trial will include patients with paroxysmal (intermittent) AFib and patients with persistent AFib. If successful, that study could lead to the Abbott Volt PFA system’s FDA approval and product launch as early as 2026, though Abbott has not yet shared a timeline for approval or commercialization.
“There are obviously risks down the road, [but] we try to be as fast as possible. … We see excitement on the enrollment side. These trials enroll really fast,” Piorkowski said. “Where we stand right now, we’ve treated 30 patients successfully in in Australia. These patients go through an intermediate assessment of safety and efficacy, and this data will be critical for the discussion of when we can start our IDE study.”
This post was originally published on Jan. 22, 2024, and updated on Jan. 31, 2024.