Technology developments in the rapidly evolving Medtech industry now make it possible to replace a “one-size-fits-all” approach to fitness and healthcare. Developers are now able to create customized solutions using biomarker sensing for more individualized monitoring of one’s physiology. Biomarkers are specific physiological indicators used to determine risk factors for conditions or illness. They can provide individuals and their physicians with important data to more quickly identify or corroborate a condition or disease that may require intervention in the form of treatment.
Physicians routinely use biomarkers to diagnose and monitor patients who may have cancer, heart disease, or previously undetectable signs of diabetes. Noninvasive biomarker sensors that accurately measure changes in sweat and other biological fluids, can allow for a more personalized—also known as a “patient-centered”—approach to fitness goals and healthcare management. Early detection of certain biomarkers can improve the efficacy of treatment plans, and reduce the incident rate and hospitalizations due to adverse events. Early detection of life threatening diseases can potentially save lives.
Figure 1. Wearable Sensor (Image Credit: Molex)
Popular wearable fitness devices detect metrics such as number of steps, heart rate, and calories burned. Today there’s growing interest in wearable devices that go beyond measuring biometric parameters to measuring biomarkers in bodily fluids. Blood, sweat, saliva, and other biofluids are widely used for clinical analysis in healthcare settings. Continuous monitoring of biomarkers via bodily fluids is painless and non-invasive, and can help assure patient compliance—all major advantages for people who are at risk for or living with chronic conditions, and helpful as well for their physicians. The successful achievement of accurate and noninvasive continuous monitoring of biomarkers would represent enormous advances for healthcare—and result in significant market opportunities for sensor device developers.
The unique characteristics of sweat allow for noninvasive rapid collection and testing when compared with blood or other biofluids. Researchers, startups, and industry leaders are actively working on projects to create and test sensors capable of measuring various biomarkers in human sweat. Several prototype devices have leveraged highly sensitive carbon nanotube sensing and developments in the fabrication of thin, flexible silicon die to achieve moderate progress. Figure 1 shows the typical make-up of a biomarker sensor.
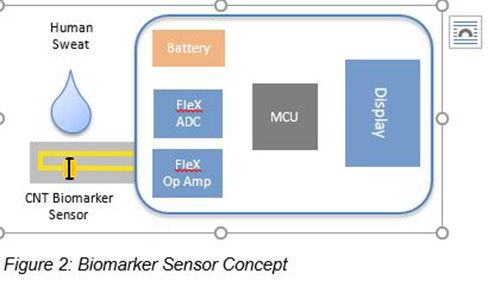
Figure 2: Biomarker Sensor Concept (Image Credit: Molex)
Lactate, for example, is a potentially important biomarker present in sweat, as it can indicate the onset of fatigue of an individual and has been indicated as a marker for all-cause mortality in hospital patients. Progress has also been made toward developing sweat-based glucose monitoring, with potential for a combination transdermal drug delivery system for multiple medications. Skin-based sensors can also test for the presence of other metabolites, such as electrolytes, calcium and heavy metal ions in sweat. Collaboration between biomedical engineers and material scientists has led to the creation of small wearable flexible sensors that are able to read multiple biomarkers in the molecular composition of sweat and transmit that data to a smart phone for real time analysis and tracking.
Both technology and manufacturing readiness level assessments for biomarker sensors are important to identify gaps and risks for their development. Many challenges exist in the development of flexible sensors capable of detecting biomarkers at concentration levels expected in sweat or other bodily fluids. Biomarkers exist in smaller concentrations in sweat and saliva than in blood samples. The time to collect a sample of adequate volume and concentration is highly individualized depending on output, ambient temperatures, and other environmental factors. Providing consistent selectivity and sensitivity of readings of a complex fluid at an affordable cost for wearable and disposable devices are significant obstacles for sensor developers and manufacturers to overcome. Biofluid samples can be constantly refreshed in a lab. Although advancements in microfluidics have been promising, real world test subjects have demonstrated them to be insufficient to replenish sweat at a sufficient rate to maintain an equilibrium. One example is in a sweat monitoring use case where sensor selectivity, sensitivity, stability, and reliability are critical.
Disposable biomarker sensors are typically functionalized using chemical compounds that act as receptors for specific biomarkers, which then turn that chemical reception into a variable resistive load on the sensor. A number of intriguing biomarker sensor technologies developed with the intention of the sensors eventually being incorporated into a wearable, disposable device have been demonstrated successfully in labs, but they will require further development to achieve desired performance metrics for demanding wearable products. Sensors must be even more sensitive and stable with lower concentrations of samples in the interest of achieving the application’s goals.
Sensor Demonstration Platforms
In the meantime, Molex has developed various thin and flexible sensor demonstration platforms that can interface with a variety of disposable sensors. The sensor platforms are typically custom configured to make a signal interpretable within the given sensor responsiveness and application requirements. Sensors in these demonstration platforms typically utilize a higher concentration than will be achievable in the ultimate wearable sensor devices to assist in showing the promise of the technology while also acknowledging its gaps. Reception of the biomarker compound effectively turns into identifiable load on the sensor, which is then typically amplified and digitized and communicated to a processor to perform logic computations and ultimately communicate to the user via an indicator display or other HMI (eg. mobile app).
Commercial success of a device depends on sensor system form factor and performance, in addition to manufacturability and ultimately, cost. The reliability of electronics integration is ahead of the commercialization curve relative to biomarker sensor technologies. A range of sensor platforms can be used to model biomarker signals on prototypes and for conducting sub-system functional testing. Key features of a typical biomarker sensor platform would include a microprocessor, an operational amplifier and an analog-to-digital converter, indicator LEDs or thin and flexible display, antenna, possibly additional memory, and coin cell or thin film style batteries.
Integrated sensor design concepts can be verified with a quick turn FR4-based printed circuit board assembly to limit variables in new designs. Recent developments in flexible functional printing and assembly materials and technologies allow for more economical, lighter weight flexible hybrid sensor electronics (Figure 2) than traditional rigid or flexible etched copper circuitry. Biomarker sensor platforms on silver flexible circuitry permit highly complex, yet flexible and durable, double-sided circuitry layouts. A typical system might comprise a polyethylene terephthalate (PET) substrate with silver and dielectric ink application using widely available printing techniques. Depending on the application requirements, an assembly can incorporate, for example, a 0.50mm pitch QFN package for the IC, encapsulation to protect critical components, zinc manganese or lithium polymer based thin film batteries, biocompatible mounting adhesives, and graphic overlays to demonstrate an aesthetically pleasing finished product.
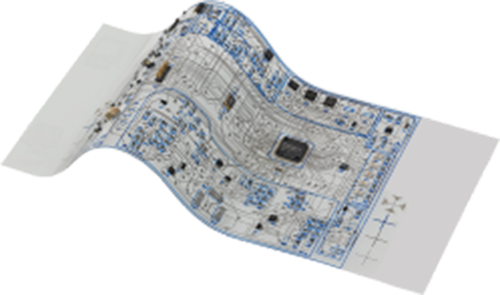
Figure 3. Flexible hybrid circuits allow the development of lightweight, fully integrated wearable sensors. (Image Credit: Molex)
At the core of an integrated sensor platform, a power budget is first considered, then firmware is developed to program microcontroller function, the microcontroller processes amplified, and digitized data from the sensor, op-amp, and analog-to-digital converter, and the data is transmitted to a smart phone or other mobile device via wireless technology, often using near-field communication (NFC 13.56MHz) or Bluetooth Low Energy (BLE). Currently, most integrated sensor designs range from 2 to 5 mm thick. A true wearable biomarker form factor needs to be thinner to be the most conformable and least intrusive to the user. Thinner silicon die will allow for a thinner and more flexible substrate. High-speed sheet-to-sheet and roll-to-roll printing and assembly technology can allow high-volume handling of much thinner substrates (1 to 2 mm thick).

Figure 4. Flexible sensor platform integrates antenna, thin flexible display, microcontroller, and thin flexible batteries. (Image Credit: Molex)
Important progress has been made to improve consistency in thin flexible sensors to detect a range of biomarkers in human biofluids. However, the requirements for wearable devices contacting biofluids are more nascent and nuanced than sensors used to count steps or measure other external factors such as vibration, shock, ambient or surface temperature and humidity. More work will be needed to build miniaturized wearable integrated biomarker sensor solutions that are reliable and accurate, disposable and economical. Pioneering developers are now working toward the vision of low cost conformable electronics that can be worn for monitoring human health and performance metrics.




