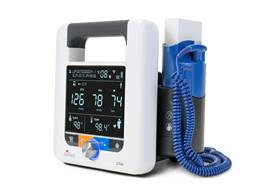
The instrument’s modular design allows clinicians to easily make adaptations to the device, enabling them to add thermometry, SpO2 and Wi-Fi while in the field and based solely on the needs of their patient.
Blood pressure technology expert SunTech Medical has received FDA approval for U.S. distribution of its next generation spot-check vital signs device: the SunTech CT40. Driven by SunTech’s clinical-grade Advantage BP technology, the new spot-check vital signs device provides accurate and reliable automated measurements of blood pressure, temperature and pulse oximetry with clinical-grade performance, advanced features and digital connectivity.
The instrument’s modular design allows clinicians to easily make adaptations to the device, enabling them to add thermometry, SpO2 and Wi-Fi while in the field and based solely on the needs of their patient. Available modules include Covidien’s FILAC 3000 thermometry and Masimo’s SET Sp02 technology.
Another feature of the device is SunTech’s Customizable BP Averaging. BP averaging is a key component to accurate blood pressure measurement and used in the recent SPRINT trial conducted by the National Institute of Health.
It is rapidly becoming the new standard of care, with improved accuracy and reduced white coat response. SunTech’s Customizable BP Averaging allows clinicians to automate the process of taking and averaging multiple office BP measurements, while setting their own averaging protocol that fits best into their practice workflow. The device also allows users to verify automated readings using a stethoscope without removing the cuff for consistent patient BP measurements.
The SunTech CT40 connects to a network through Ethernet or optional Wi-Fi, and it uses industry standard IHE profiles to facilitate communication of patient information and measurement data with the EMR system. The device’s built-in memory can store up to 99 vital signs charting sessions and users can also easily document patient’s measurements electronically to avoid transcription errors.
A robust mobile stand is available to facilitate room-to-room relocation. A barcode reader option is available for easy data entry, as well as a thermal printer for hard copies of vital signs measurements.
SunTech Medical
suntechmed.com


![A photo of the Medtronic GI Genius ColonPro polyp detection system flagging a potential sign of colon cancer during a colonoscopy. [Photo courtesy of Medtronic]](https://www.medicaldesignandoutsourcing.com/wp-content/uploads/2024/04/Medtronic-GI-Genius-doctors-268x170.jpg)

