Moving diagnostics out of the lab and putting it right at the patient’s bedside or even into the patient’s hands is the goal of many medical technology companies. The advantages that can be realized from the rapid diagnosis of disease or some other health concern are numerous. Additionally, the effort to better equip patients to manage their own health is a priority for many.
With this in mind, I interviewed Dr. Dale Athey, CEO of OJ-Bio, a company that is developing a point-of-care, connected, mobile diagnostic device that accomplishes many of the aforementioned goals. In the following presentation, Dr. Athey explains the device, provides insights on design challenges, and speaks to diagnostic technology trends that are relevant now and in the future.
Sean Fenske: Thank you for speaking with MDT. Let’s start with your device. Can you please tell me about your diagnostic technology solution and what it does?
Dr. Dale Athey: OJ-Bio is a joint venture company that has developed new technology capable of measuring diagnostic biomarkers on special biochips by combining the technologies of its two parent companies, Japan Radio Company (JRC) and Orla Protein Technologies.
The Xtalline technology developed by OJ Bio represents the state-of-the-art in mobile-enabled medical diagnostic testing and patient monitoring. The biosensors used in Xtalline are converted surface acoustic wave (SAW) chips, which are normally used in mobile phones and other electronic devices. The biosensor chips developed by the joint venture combines the specialist nano-protein surfaces developed by Orla to detect the presence of disease, which is converted into an electronic signal by the specialist JRC SAW chips. The biochip is inserted into a reading device and the presence of the resultant electronic signal can be used to diagnose and indicate the presence of the infectious disease being tested for, which is displayed on a complementary smartphone app or PC-based software program.
When biological samples from serum, urine, saliva, or blood are applied to the biochip (held in a disposable cartridge), the presence of a disease antigen causes a shift in the phase angle of the surface acoustic wave passing across the chip surface. This is translated into an electronic signal. This signal can then be detected and its presence (or absence) determined, providing an unequivocal pass/fail result for the particular disease being tested for. Unlike other rapid test systems, this can provide a measure of the target biomarker, rather than just a simple yes or no result. This is particularly useful in monitoring the disease and evaluating the effectiveness of the treatment.
The Xtalline platform technology developed by OJ Bio allows the detection of different protein biomarkers, meaning the potential application areas are very broad. Our lead application is a test for C-Reactive Protein (CRP), which is a recognized biomarker of inflammatory disease and can be used to rule out serious bacterial infections. Although the CRP test is not new, its availability on a point-of-care diagnostic device, potentially for use by patients themselves as well as frontline healthcare staff, could provide a useful tool in the reduction of the inappropriate prescribing of antibiotics for flu-related conditions.
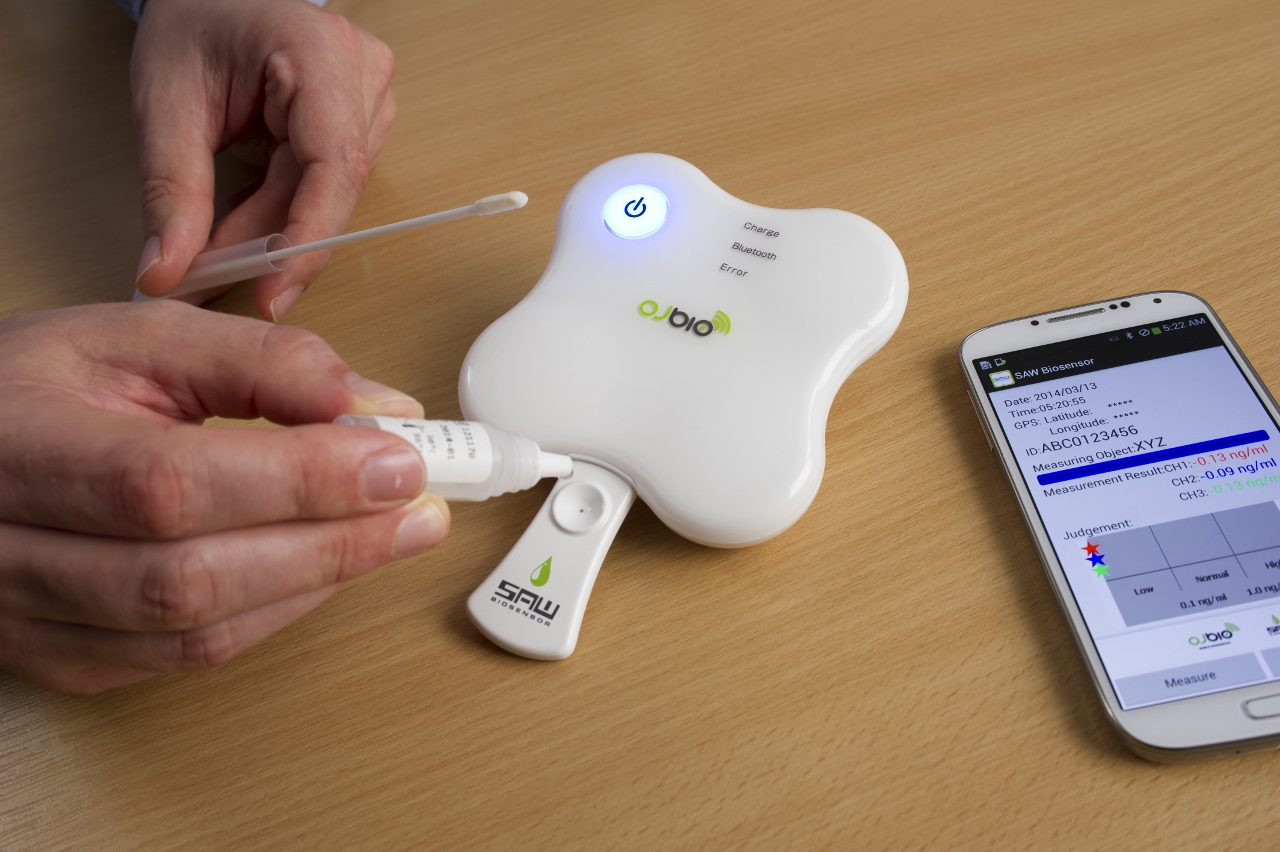
The Xtalline platform technology developed by OJ Bio allows the detection of different protein biomarkers at the point-of-care.
Fenske: Why is it important to reduce the inappropriate prescribing of antibiotics?
Dr. Athey: Antibiotics are needed to treat diseases caused by bacteria. Over recent decades, antibiotics have often been prescribed inappropriately for infections caused not by bacteria but by viruses, and often because GPs are under pressure to make an immediate diagnosis and start treatment. Getting test results back from a laboratory can take a long time and requires the patient to come back to the GP’s office or some form of follow-up contact.
This has resulted in the inappropriate prescribing of antibiotics and has fostered growing antimicrobial resistance, rendering the antibiotics ineffective for some treatments. To protect antibiotics for the future and to decrease the costs associated with the drug-resistance, it is of great importance to prescribe antibiotics only when they are necessary.
Having an effective point-of-care diagnostic device available can help GPs and other prescribers demonstrate to patients why antibiotics may not be appropriate and reduce unnecessary use.
Fenske: How are point-of-care diagnostic technologies changing healthcare?
Dr. Athey: Point-of-care testing devices provide very quick and effective diagnosis of a disease or infection. This is important for some diseases and can also reduce the cost of traditional processes associated with laboratory testing.
For example, testing for CRP in laboratories is possible but not adopted very well by GPs and other professionals as patients often request immediate results. Studies have showed that point-of-care tests for CRP reduced antibiotic prescription by 31 %, which is a significant step in fighting growing immunity to some antibiotics.
Importantly, the diagnostic device can be used at a patient’s bedside, in a pharmacy, or even in the patient’s home with the results being available immediately and without samples being sent for laboratory analysis. This will enable portable testing to be undertaken by patients themselves or nurses. Xtalline is very simple to use and has low requirements for training or maintenance
Fenske: What were the major challenges of developing this technology?
Dr. Athey: Our technology combines electronics and biology, which is a major challenge. OJ-Bio is a joint venture company created by Japan Radio Company, a multinational Japanese company with expertise in wireless communication, and Orla Protein Technologies, focused on nanobiotechnology. Combining the technologies was challenging and required a lot of collaboration and investment, but the result is very innovative. The huge interest by companies in the life science sector confirms we are on the right track.
Our work shows that Xtalline has huge potential, and we believe we can be leaders in the convergence of mobile, bio, and electronic technologies in healthcare.
Fenske: How were you able to “shrink” processes that required a laboratory to complete into a handheld device?
Dr. Athey: Through application of our respective expertise in nanobiotechnology (working at the nanoscale with precise biological recognition surfaces) and state-of-the-art electronics from our parent company in Japan. The combined expertise with a clear goal in mind has resulted in the technology as it stands today.
There will always be a need for definitive, laboratory-based molecular testing methods capable of screening for all types of infectious agents. However, effective testing and diagnosis using portable devices has considerable potential in community screening programs.
Overall, the ability of point-of-care testing to deliver a fast and accurate result will have significant benefits in certain situations — and particularly in those situations where fast diagnosis is essential. For example, if a patient presents with a suspected condition at a healthcare facility during “out of hours” periods, the point-of-care device would enable an immediate diagnosis to be made, allowing clinical decisions to occur quickly with laboratory verification or confirmation testing being available later.
.jpg)
Dr. Dale Athey is the CEO of OJ-Bio.
Fenske: Is this technology able to be adapted to address other similar point-of-care diagnostic procedures?
Dr. Athey: Yes. As mentioned previously, Xtalline is very adaptable and flexible. CRP is our lead application and will be fully developed in 2016, but we have others in the pipeline. For example, considerable work has already been undertaken on the use of the specialist biochips for the detection of flu-like respiratory diseases and periodontal gum disease. In other areas of infectious disease identification and control, OJ Bio has worked with researchers from University College London on the development of a point-of-care device capable of detecting HIV marker proteins in human blood quickly and effectively.
In principle, Xtalline technology could measure any kind of biomarker incorporated on our biochip.
Fenske: What is the value of making this device a connected technology?
Dr. Athey: The impact of mobile technology on healthcare is enormous and growing at a rapid rate. The connectivity to smart phones and healthcare systems opens up great opportunities. Smartphones are used more and more for health applications. Having the test results on the phone makes it incredibly easy to get test results and to monitor them over time.
Importantly, as well as results from the biosensor device being displayed on a complementary, app-enabled, handheld reading device such as a mobile phone, the data can also be wirelessly transmitted to a central resource for remote connectivity to healthcare networks, records, and facilities. This capability also makes the technology ideal for remote disease monitoring, with many applications for rural use or situations where clinical readings are needed without patient transfer.
Collating records from different screening centers also means it could be used to monitor the spread of diseases and thereby help healthcare professionals react quickly in cases of epidemics or other events.
The use of this technology is also part of the wider democratization of healthcare — empowering patients and consumers to take further control of their own health.
Fenske: Why did you make a smartphone serve as the readout mechanism rather than bundling it into the device itself?
Dr. Athey: There are still regulatory issues around the use of the phone in healthcare applications. If the device is bundled into the phone, then it becomes a medical device itself and that may slow the regulatory path and adoption.
We think that this approach is best at the moment in that it will speed approval and adoption and get the technology out to those who need it faster. In the future, there will be bundling of the type you describe.
Fenske: Are there concerns with data security with this device? How did you address them?
Dr. Athey: Wherever medical data is concerned, we must have the highest regard for security and confidentiality. However, we don’t see this as a barrier and feel that the use of the device with the correct regard for security will improve on the current situation where people often do not have access to their own health data or they may be using insecure means of recording sensitive information such as writing results in a journal or similar.
Fenske: What’s on the horizon for this technology?
Dr. Athey: Xtalline-CRP will be ready by the end of 2016 and the biochips for HIV, flu, and gum disease are also in the pipeline for further development. We are continuously innovating for developing more application areas, and we are also looking for partners for codevelopment or comarketing agreements to bring the Xtalline technology to market.
Fenske: Before concluding, do you have any comments you’d like to share?
Dr. Athey: The convergence of technology, bio, and mobile for healthcare applications is now moving forward at a rapid rate. This is moving testing and monitoring closer to the patient and empowering both healthcare workers and consumers to make more decisions in a timely fashion.
New business models are emerging and the healthcare landscape is undergoing a huge transformation. This transformation will involve cooperation between all of the key stakeholders: healthcare providers, pharma, medical devices and diagnostics, and IT and technology companies. OJ-Bio intends to be a key player in this revolution.




