Transit Scientific this week announced that it has received FDA clearance for its hydrophilic-coated XO Cross microcatheters for guidewire support, exchange and contrast media injection in the peripheral vasculature.
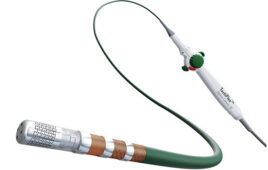
Transit Scientific this week announced that it has received FDA clearance for its hydrophilic-coated XO Cross microcatheters for guidewire support, exchange and contrast media injection in the peripheral vasculature.
The 510(k) clearance includes new 2Fr XO Cross 14 microcatheters, 2.6Fr XO Cross 18 microcatheters and 3.8Fr XO Cross 35 support catheters. They can be used with standard 0.014 in., 0.018 in. and 0.035 in. guidewires in 90 cm, 135 cm, 150 cm and 175 cm working lengths.
Park City, Utah-based Transit Scientific designed the XO Cross platform with a metal-alloy exoskeleton, non-tapered shaft, and proprietary hydrophilic lubricious coating. It is designed for use in peripheral lesions but not limited to above-the-knee and below-the-knee CTOs from contralateral femoral, antegrade femoral and retrograde pedal access.