Alzeca Biosciences announced successful preliminary results for ADx, its proprietary diagnostic imaging technology, designed to enable diagnosis of Alzheimer’s Disease for the first time on conventional Magnetic Resonance Imaging (MRI), providing results a decade or more before the onset of symptoms of cognitive decline.
Successful development of ADx would make early and reliable Alzheimer’s testing available to millions, using scanning technology that is widely available, and at far lower cost and without the radiation of current Positron Emission Tomography (PET) scans. Earlier detection of Alzheimer’s could vastly improve the efficiency of clinical trials for new therapies by identifying appropriate candidates for participation.
“Alzeca is building a pipeline with a multi-modality and multi-disease approach using our proprietary technology platform ADx,” said Carlo Medici, Alzeca Biosciences CEO. “With the first positive results, we will now aggressively bring this unique technology to the next phases of development and pursue partnerships that will enable us accelerate the company’s strategy.”
Alzeca also is exploring the significant additional potential of the underlying platform of ADx, which can be modified to bind to different targets in the brain. With these modifications, the platform could be used to image and diagnose other serious neurodegenerative diseases, and ultimately be used as a drug delivery system for treating those conditions.
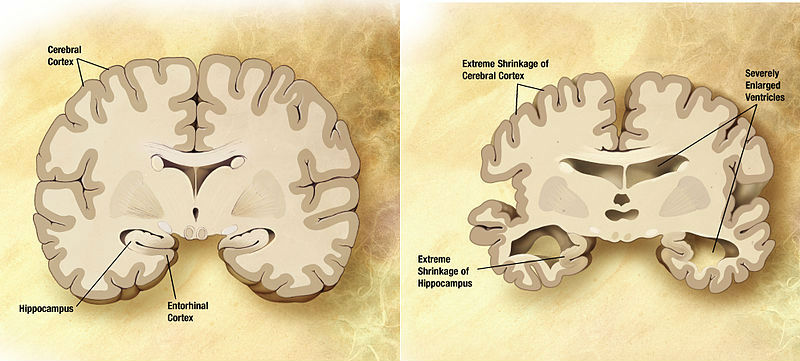
Combination of two brain diagrams in one for comparison. In the left normal brain, in the right brain of a person with Alzheimer’s disease.
Initial Test Results
In peer-reviewed studies, the ADx nanoparticle demonstrated the ability to carry a contrast agent for MR imaging with a targeted ligand across the blood-brain barrier to bind precisely to amyloid plaques in the brains of test mice. Researchers were then able to demonstrate those plaques using ordinary MR equipment. The results, along with successful preliminary toxicity studies, allow Alzeca to accelerate the development and testing of ADx, with the goal of beginning human clinical trials in 2018. In May, the company was awarded a $322,000 grant from the Alzheimer’s Drug Discovery Foundation (ADDF) to fund the next phase of testing of ADx in canine subjects.
Unmet Need
More than 27 million people suffer from Alzheimer’s disease today, and it is the sixth leading cause of death in the U.S. By 2050, the disease could result in a $1.1 trillion annual cost to the U.S. healthcare system. Yet there is no cost-effective, readily available, reliable way to diagnose the disease before the onset of cognitive decline–which is when current Alzheimer’s therapies have traditionally been most effective.
ADx would not only confirm an Alzheimer’s diagnosis years before significant clinical impairment, but also allow physicians to rule out such a diagnosis, to ensure better management for aging patients. Misdiagnosed dementia patients not only receive the wrong therapies, but the cost of treating a misdiagnosed patient adds $12,000 to the annual cost of treatment.
Significant Technology
Alzeca’s biodiagnostic platform is a nanoparticle that can carry an imaging marker or other “payload” and pass through the normally impenetrable blood-brain barrier. The outside of each nanoparticle is designed to bind precisely to specific target substances in the brain, delivering the payload to that target.
ADx carries an MR imaging agent as its payload and through a ligand targets the amyloid plaques that are the hallmark of the onset of Alzheimer’s disease, developing 10 or more years before cognitive impairment. Once ADx is bound to the plaques, beta-amyloid, the hallmark of the disease in its earliest stages, can be imaged in high resolution using a standard MRI scan for the first time.
Superior Modalities
MR scans are radiation-free, far less costly and more readily available throughout the world compared with the only current alternative, a PET scan. PET scans cost two-to-five times MRI scans and require radioactive imaging agents that preclude routine use, while ADx would permit physicians to track the patient’s condition over time and even screen for the disease.
Scientific Expertise
“We must greatly improve the diagnosis of the disease, as more than 95 percent of new treatments fail to be FDA-approved, frequently because patients enrolled in drug trials never had the right diagnosis,” Scott W. Atlas, MD, chairman of Alzeca Bioscience and former chief of neuroradiology at Stanford University Medical Center. “Early diagnosis would facilitate pre-emptive treatment, when the disease is far more likely to respond and before patients are seriously impaired. Even in advance of effective therapy, earlier detection of Alzheimer’s Disease would provide crucial information to patients and their families valuable for future financial and life planning.”




