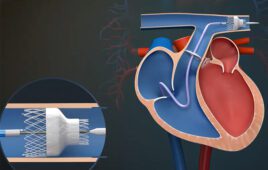
Bard originally developed the ePTFE encapsulation services its own branded stents. Bard OEM customers will now have access to the material and processes needed for encapsulating their own component or device. Such ePTFE encapsulation is especially suited for implantable medical devices such as transcatheter heart valves that are in need of the material’s thin, high strength, micro-porous, and biocompatible properties.
The latest offering comes at the same time the company’s Bard Peripheral Vascular OEM Products division celebrates its 40th anniversary. The division offers a wide range of products and services to OEM customers, including implantable textiles, implantable ePTFE, and PTA and PTCA angioplasty balloon catheters.
“For 40 years, we have been proud to provide our esteemed customers with the component products, technical support and regulatory information they need to develop the highest quality medical devices for cardiovascular applications,” Bill Gemma, director of OEM sales and marketing at Bard PV OEM, said in a news release.
Bard since the 1970s has offered a full line of more than 20 implantable textile styles, many of which are still used in devices including heart valves and left ventricle assist devices (LVADs). In the decades since, Bard PV OEM has expanded its offerings to include PTFE and PET felts, as well as ePTFE for use in advanced cardiovascular device components.
Expanded polytetrafluoroethylene is an attractive material for implantable medical devices because it is chemically inert, hydrophobic, and gas permeable. It exhibits conformability and high temperature stability.
[Want to stay more on top of MDO content? Subscribe to our weekly e-newsletter.]