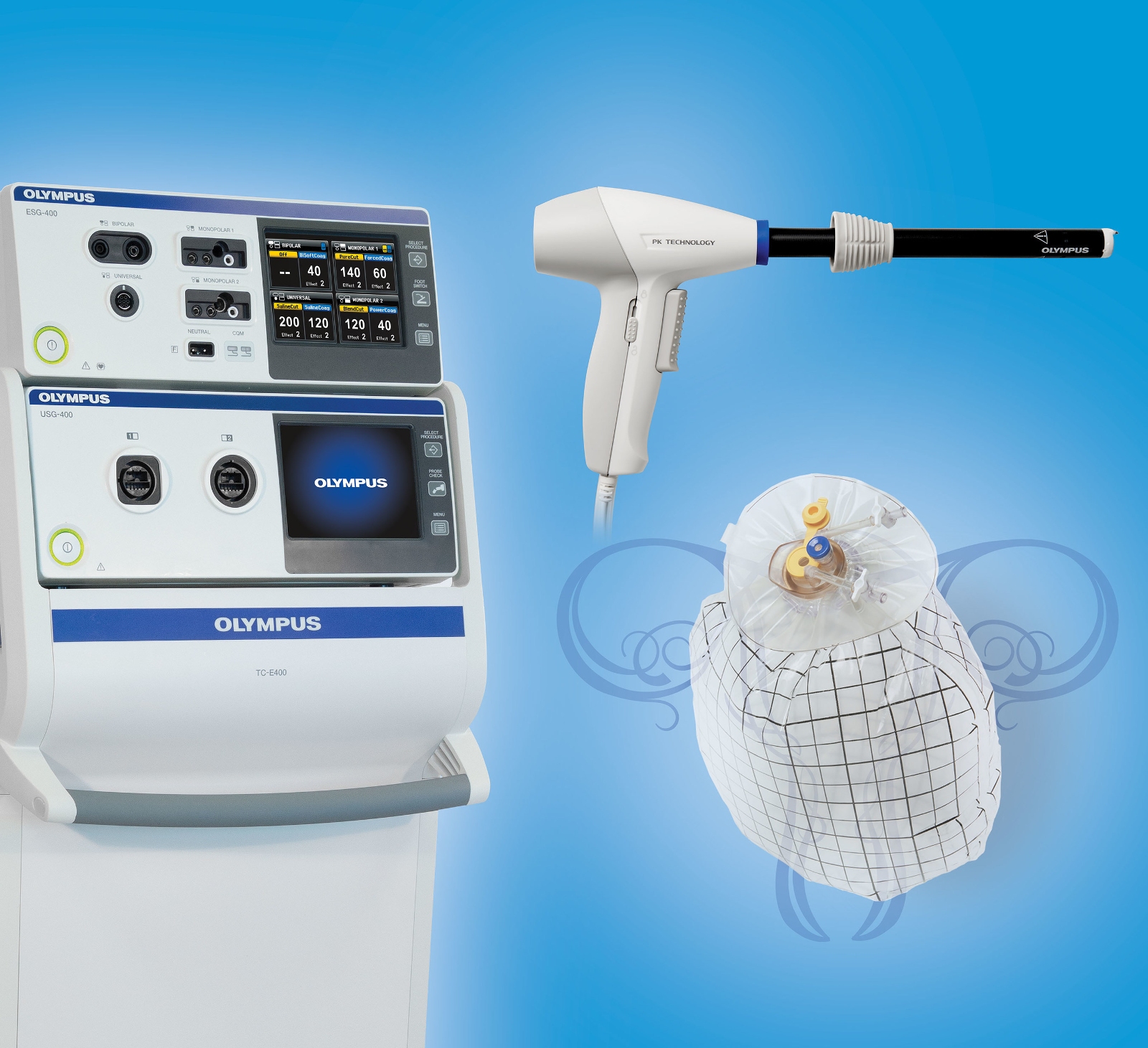
Olympus America Inc. has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the company’s next-generation laparoscopic PK Morcellator, paving the way for a complete solution for laparoscopic tissue containment and extraction.
The Olympus Contained Tissue Extraction System comprises the PneumoLiner, a first-of-its-kind containment device cleared by the FDA for gynecologic surgery in April 2016 and the next-generation laparoscopic PK Morcellator to provide certain patients a laparoscopic surgery option to avoid open hysterectomy and myomectomy for uterine fibroid removal. The system was officially introduced November 15 at the American Association of Gynecologic Laparoscopists (AAGL) Annual Meeting.
The PneumoLiner containment device and PK Morcellator are not indicated for use in women with tissue that is known or suspected to contain malignancy; and should not be used for removal of uterine tissue containing suspected fibroids in patients who are peri- or post-menopausal, or candidates for en bloc tissue removal vaginally or via mini-laparotomy.
Gynecologic professional societies—including the American Congress of Obstetricians and Gynecologists (ACOG), the Society of Gynecologic Oncology (SGO) and AAGL — have openly voiced concerns about exposing the majority of women who have benign fibroids disease to the risk of complications associated with open procedures. The gynecologic professional societies have indicated that morcellation should not be entirely removed as a treatment method and that contained tissue extraction may be a solution.
“Contained tissue extraction is continually evolving,” notes Arnold P. Advincula, MD, vice-chair & chief of gynecology at Sloane Women’s Hospital, Columbia University Medical Center/New York-Presbyterian Hospital. “As surgeons, it is our duty to evaluate potential solutions to the surgical challenges we face in clinical practice. We are pleased that Olympus has taken the initiative to develop a unique and well tested system designed specifically to facilitate the option for a laparoscopic approach in women who are at a low risk of having an unexpected malignancy at the time of surgery.”
(Image credit: Olympus)
The Olympus Contained Tissue Extraction System is the first complete solution designed for contained tissue extraction to enter the U.S. market, enabling surgeons to isolate and perform uterine tissue morcellation and extraction using the PneumoLiner containment device. The PneumoLiner is currently the first and only containment device to receive FDA market approval for use with certain laparoscopic power morcellators to isolate uterine tissue that is not suspected to contain cancer. Once insufflated, the PneumoLiner allows space and vision throughout the power morcellation procedure while maintaining a barrier to the escape of fluids, cells, and tissue fragments.
When insufflated, the PneumoLiner creates a barrier between the targeted tissue and non-targeted abdominal contents, to minimize the risk of inadvertent damage to adjacent structures. When used as directed, the PneumoLiner significantly reduces the risk of disseminating cells and tissue into the peritoneal cavity during power morcellation.
Uterine fibroids are the reason for 30-40 percent of the nearly 600,000 hysterectomies performed in the U.S. each year. The percentage of these hysterectomies performed using minimally-invasive surgery (MIS) has increased from approximately 30 percent in 2002 to about 63 percent in 2012, the benefits of which have been widely documented in retrospective and prospective studies, and include significant improvements in morbidity and mortality compared with open surgery. Myomectomy, which is the surgical removal of fibroids while leaving the uterus intact, has been estimated to be performed laparoscopically one-third of the time. Prior to its 2014 warning, the FDA estimated that approximately 60,000 morcellations were performed each year.
A strict surgeon training protocol is required and has been validated involving surgeons with varying levels of experience. Testing conducted with the surgeon training protocol revealed there was no damage to any of the specimen containment bags utilized during this process. Consistent with the FDA’s mandate, surgeons who wish to use the containment system will be required to complete a formal training protocol before use.
The Olympus Contained Tissue Extraction System for removal of uterine tissue in select patients can address key healthcare objectives:
- Increased Quality Care—The use of a laparoscopic surgical approach can increase the quality of patient care, as an open hysterectomy can lead to an increase in major surgical complications (2.2% to 2.8%) with an increase in hospital readmission within 30 days (3.4% to 4.2%).
- Decreased Costs—An increase in quality care leads to a decrease in overall patient costs, as the median episode cost for readmission was noted in one study to be $5,847, with a range of $5,478-$10,389.4 A strategy of minimally invasive laparoscopic hysterectomy is a cost effective surgical method versus an open hysterectomy, $20,853.15 vs. $30,359.92, respectively.
- Enhanced Patient Satisfaction—Patients whose surgeons use a minimally invasive surgical approach may experience fewer perioperative complications, shorter hospital stays, less pain, improved quality of life and a faster return to normal activity. A 2016 study by Po et al revealed that 65% of women, after weighing the risks and benefits, would choose a minimally invasive surgical approach if the risk of cancer spreading was calculated at 1 in 350, the estimate the FDA evaluated.
“The Olympus Contained Tissue Extraction System provides gynecologists and their appropriate, low-risk patients with what they want again: a choice,” says Todd Usen, president of the Olympus Medical Systems Group at Olympus Corporation of the Americas. “The Olympus Contained Tissue Extraction System is the solution that will allow gynecologists to once again safely and effectively offer minimally invasive hysterectomies and myomectomies as options to certain low-risk patients.”




