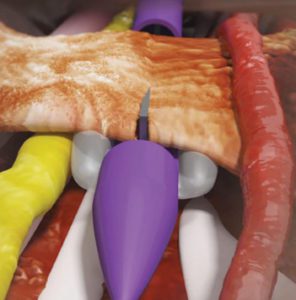
The Stealth MicroKnife (the thin grey wedge) is positioned while sheathed. Other device features protect surrounding nerves and vessels.
Carpal tunnel syndrome affects more than 12 million Americans and results in 500,000 surgeries each year. The traditional fix has been an invasive surgery that involves cutting the transverse carpal ligament in the wrist, which is not expansible. The ligament forms the roof of the carpal tunnel and can pinch a nerve in the wrist causing the discomfort experienced in carpal tunnel syndrome patients. The conventional solutions expand the space around the nerve.
The ultra-low-profile Stealth MicroKnife device uses technology developed at the Mayo Clinic to address carpal tunnel syndrome. The first MicroKnife prototypes were able to reduce the size of the traditional incision from two in. to about ½ in. (12.7 mm). A few prototypes later, the device now requires an incision of 3 mm to 5 mm.
Once properly positioned using ultrasound, the device provides a “safe zone” barrier to protect nerves, blood vessels, tendons and other sensitive anatomic structures during transection. The instrument will let physicians perform carpal-tunnel-release surgery through a single micro-incision with ultrasound guidance (or with a traditional mini-open incision without ultrasound guidance) while protecting sensitive anatomy during transection of the transverse carpal ligament. MicroKnife requires an incision so small it can be closed with a small adhesive bandage or strip instead of sutures.
Immobilization is unnecessary, so patients can begin rehabilitation and get back to their jobs and lives almost immediately. The result is a low-trauma outpatient procedure associated with a recovery that is shorter than that of conventional surgical methods.
The procedure formerly took place in an operating room but the innovation will let physicians safely and effectively perform carpal tunnel release in an office or ambulatory surgery center. This results in rapid patient recovery, improved cosmesis and reduced costs.
The company was founded in 2014 by Barnes and Smith, both physicians at Mayo Clinic, and business operations expert Aaron Keenan. Regarding funding, Barnes and Keenan, who both serve as CFO, would only say the private company raised sufficient capital by July 2015 and is funded to reach key milestones, such as inpatient use.
The Stealth MicroKnife is not for sale yet. Prototypes have been successfully tested on cadavers multiple times and gone through several iterations of testing and redesign. The first human patient is scheduled to undergo the procedure during the fall of this year.
Sonex Health
sonexhealth.com




