 1. Medtronic
1. Medtronic
R&D Spending: $2,331,000,000
Percentage of revenue: 8.1%
2020 overall ranking: 1
In fiscal year 2020, one of Medtronic’s biggest accomplishments was publishing open-source design specifications for its Puritan Bennett 560 portable compact ventilator in March 2020 when the COVID-19 pandemic was just getting started. The company said that in the first few weeks of the open-source design being available, there were more than 200,000 downloads of the Puritan Bennett 560 blueprint. The ventilator also received FDA emergency use authorization for use in COVID-19 patients, and later launched a remote management capability to allow clinicians to adjust ventilator settings from outside a patient’s ICU room.
Medtronic also touted diabetes, TAVR and other cardiac rhythm and neurostimulator accomplishments in its most recently reported fiscal year.
In June 2019, Medtronic started a pivotal trial of its next-generation Guardian continuous glucose monitoring sensor. The trial studied the sensor in up to 460 patients with type 1 or type 2 diabetes aged two to 80 years old over the course of seven days. In the same month, Medtronic’s diabetes business also enrolled the first study participants in its pivotal trial of the Bluetooth enabled MiniMed 780G advanced hybrid closed loop system that is designed to automate the delivery of correction boluses.
Medtronic also received an expanded TAVR indication in August 2019. The indication was extended to symptomatic severe aortic stenosis patients at low risk for surgical mortality.
In September 2019, the company launched its next-generation Evolut Pro+ TAVR system after it received FDA approval. The system features four valve sizes with an external pericardial tissue wrap that provides advanced sealing for the largest indicated patient treatment range and a low delivery platform.
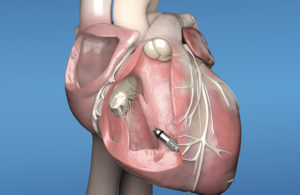
Medtronic’s Micra AV leadless pacemaker has the same size and shape of the original Micra but includes new internal atrial sensing algorithms. [Image courtesy of Medtronic]
Also in January 2020, Medtronic’s Percept PC neurostimulator won CE mark approval and launched in Europe. The technology senses and records brain signals while delivering therapy to patients with neurologic disorders.
In the fiscal year 2021, Medtronic has so far spent $5.2 million on research and development, according to its most recent SEC filing.
Some of Medtronic’s accomplishments in its current fiscal year include approvals for some of its diabetes products. It received CE mark approval for its MiniMed 780G insulin pump in June. The company’s MiniMed 770G insulin pump received FDA approval in August for patients with Type 1 diabetes aged 2 to 6 years old. The company also launched its ‘smart’ insulin pen, InPen, in November.




