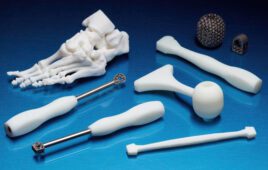
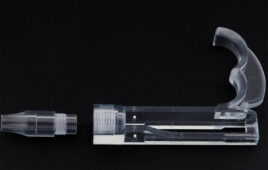
Si-Bone said today it won FDA 510(k) clearance for its 3D-printed titanium iFuse-3D implant designed for fusion procedures to treat certain disorders of the sacroiliac joint.
The San Jose, Calif.–based company said that the iFuse-3D is the 1st ever 3D-printed titanium implant designed for use in the sacroiliac joint. The company developed it using proprietary 3D printing technology that it claims has an enhanced porous surface which resembles the trabecular structure of cancellous bone.
“The design and development of the iFuse-3D implant was a multi-year effort. Our goal was to expand the iFuse family using 3D-printing technology to provide enhanced surface characteristics while retaining key performance features of the iFuse Implant, including superior rotational resistance, mechanical strength and ease of use with our existing instrumentation. iFuse-3D, with its trabecular-like surface, provides 250% greater surface area than our highly successful iFuse Implant. Additionally, the structural fenestrations allow complete bone through growth,” CTO Scott Yerby said in a prepared statement.
Si-Bone said that the iFuse-3D’s implant surface and structure were specifically designed to provide “enhanced osteointegration and promote intra-articular fusion,” according to a press release.
The company said that a study published this month in the International Journal of Spine Surgery showed significant bone growth on and into the implant’s porous surface and through its fenestrations.
“The iFuse-3D Implant embodies our commitment to advancing the diagnosis and treatment of chronic SI joint disorders through innovative products, procedures and educational programs in order to help patients suffering from chronic SI joint pain. Our team of almost 200 employees is incredibly excited to support the more than 1,300 surgeons who have used the iFuse Implant, and we believe that expanding the iFuse family has the potential to help thousands more patients. What can be more exciting and rewarding than that?” CEO & prez Jeffrey Dunn said in a press release.
[Want to stay more on top of MDO content? Subscribe to our weekly e-newsletter.]